(TiO2 induced “yellowing/pinking” with additional comments on discoloration associated with “over-oxidation” of phenolic antioxidants.)
Overview of “yellowing/pinking”
Yellowing and pinking are most often, but not always, two versions of the same problem— discoloration associated with the over-oxidation of a phenolic compound. The discoloration associated with the over-oxidation of a phenol can usually be sorted into two categories: 1) an insufficient amount or incorrect selection of antioxidant necessary to adequately stabilize the polymer under “normal” (or extreme) processing conditions; and/or 2) chemical interactions that oxidatively transform a phenolic antioxidant under conditions which are unrelated to melt processing or other sources of thermal abuse.
By far, the most common source of phenol present in polyolefins is the phenolic antioxidant (AO), typically added to the polymer to ensure good melt processing stability and some degree of long-term thermal stability. The role of phenol based antioxidants is to donate hydrogen atoms to quench free radicals, as well as to generate phenoxy groups which can also scavenge free radicals. Their fate is to serve as sacrificial reagents, which are consumed in order to protect the polymer during it’s life cycle and/or repeated heat histories. Phenolic antioxidants, as sacrificial stabilizers, can however, be over-consumed or over-oxidized if the processing conditions are too severe. This over-oxidation can lead to the formation of colored species which will be described further below.
Depending upon the heat history (temperature and dwell time), “extreme” processing conditions may consume (over-oxidize) the level of phenolic AO available for protecting the polymer from degradation. AO selection, concentration, and whether or not the phenol is combined with a secondary phosphite have an impact on the phenol-based, primary AO’s tendency to be consumed to the point of being over-oxidized.
The mechanism of the stepwise oxidation of BHT (as a simple example of a phenolic antioxidant) is shown in Figure 1. The most common product of the over-oxidation of phenol based antioxidants are quinones, which in dilute concentrations range in color from bright yellow to yellowish tan, based on the structure of the original phenolic antioxidant.
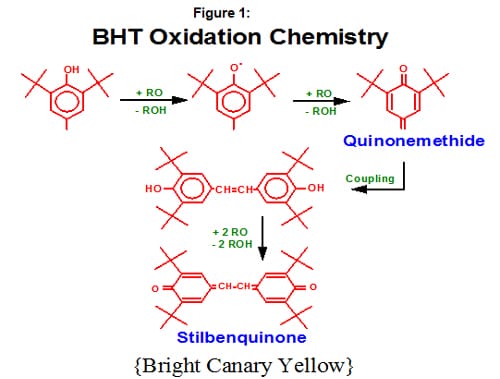
Other types of phenols can also be found in certain types of cardboard, which can result in another type of discoloration phenomenon called “cardboard yellowing.” Still, another source of phenol comes from the undesirable decomposition of hydrolytically unstable phosphites. (Phosphites are often used as a secondary antioxidant in combination with the phenol-based primary antioxidant. Some phosphites are intrinsically more hydrolytically stable than others.)
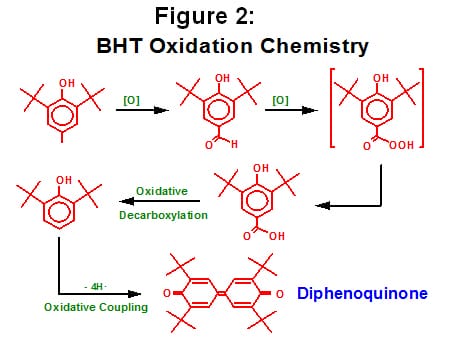
Many users of polypropylene refer to yellowing (in a warehouse situation) as “gas fading” (or refer to certain AO’s and resins as possessing good “gas fade resistance”). This is due to the fact that phenolic AO can over-oxidize by reacting with the free radicals found in an atmosphere that is polluted. For example, phenolic antioxidants are known to discolor in the presence of gaseous, combustion products containing oxides of nitrogen (e.g. nitrogen dioxide and nitrous oxide from forklift traffic, a gas or oil fired heater, or even ambient air pollutions in regions of the world), resulting in the formation of quinones. See Figure 2.
Quinones, as a pure compound, are often bright red chromophores that can appear anywhere from yellow to pink, based on their concentration as well as the presence of other additives in the system. Typically, over-oxidized phenols give a yellow cast, in a non-pigmented polymer matrix. However, in the presence of other co-additives, this yellow cast can be changed to a pink color, due to chemical transformations of the yellow color body. A representative example of pinking can be seen with over-oxidized phenols in the presence of certain types of alkaline (basic) hindered amines. A separate, but different representative example of pinking can be seen with over-oxidized phenols in the presence of certain types of TiO2.
(Note: Most forms of discoloration are relatively harmless to the polyolefin, and some people consider the chemistry to be completely reversible. The reversibility of the discoloration chemistry has been “demonstrated” by placing the discolored plastic in UV-rich light (e.g. sunlight), which “bleaches” out the color by causing the quinone to go “away;” however, this reaction is not truly reversible. In reality, the UV light is only transforming the chomophore to another structure that is much less colored. For the reaction to be considered reversible, the over-oxidized phenol would have to re-collect the same number of hydrogen atoms it donated during the polymer stabilization reactions, in order to be chemically reduced back to a phenol. UV light is only electromagnetic radiation and is not a source of hydrogen atoms.
Yellowing in an oven is much the same phenomenon, but in the absence of nitrogen compounds, the discoloration shows primarily as yellow. Zn-stearate has been shown to help here as well, by forming colorless species rather than the yellow chromophores. Other types of compounds commonly referred to as acid scavengers can also have a surprising positive or negative impact on the “color” of the system. The amount of discoloration has been found to be sensitive to the selection of the phenolic antioxidant.
TiO2 Types vs. “yellowing/pinking”
This discoloration phenomenon can be aggravated by some grades of TiO2. Non-durable rutile is most likely to contribute to this problem, due to its low level of end-treatment (which is what also makes it a terrific grade of TiO2 for applications in excess of 550o F). The low treatment level encourages the formation of titanium-based quinone complexes (which are red).
The most effective way to prevent these complexes from occurring is to make another transition metal complex available to the phenol that results in an uncolored metal-quinone complex. Certain compounds based on zinc ions have been found to be useful, since many Zn-based quinones have been shown to be colorless. Zinc stearate offers many ions in a state suitable for this reaction to occur.
In Ampacet’s technical opinion, only a few TiO2’s are suitable for extrusion processes exceeding 525 deg. F. Many other grades of “uncoated” rutile exist which may be borderline-acceptable for such high temperature applications. (If stressed enough, these borderline grades can exhibit “lensing” in high-heat, high-shear situations due to volatilization of their surface treatments or other materials adsorbed onto the TiO2 particle surface.)
The general rule-of-thumb is that the lower the treat level on the TiO2, the more suitable it is for higher temperatures. As stated earlier, though, the lower the treat level, the more likely it is that the TiO2 can “catalyze” phenolic discoloration.
In addition, most of the highly treated grades of TiO2 (“weathering grades”) also exhibit good resistance to promoting phenolic discoloration, as the surface of the TiO2 is unavailable for chemical reactions. Unfortunately, these gradesare poor candidates for most processing applications that exceed 450 deg. F.
Additional Influences on “yellowing/pinking”
Other factors that can aggravate the phenol-to-quinone discoloration reaction are darkness, moisture and alkalinity, or compounds which have high pH. (The term pKa is sometimes used here as it more correctly describes acid/base properties in a material that does not exist as a solution.)
The presence of certain types of UV inhibitors (UVI’s) can alter the pH. The higher the pH, the greater the tendency to see phenolic discoloration, especially “pinking.”
Put another way, discoloration (pink, yellow, peach and even turquoise) results from the presence of over-oxidized phenols. TiO2 types, darkness, moisture, higher pH, and NOx gases can aggravate or minimize this inevitable reaction. It is important to recognize that most people consider any type of discoloration undesirable; however, the discoloration is only a manifestation of a improperly formulated product. The root cause of the discoloration is really the subject of concern. That is why it is important to understand that if discoloration does occur, it is a signal that some aspect of the polymer composition, or the processing conditions leading to the discoloration, needs to be addressed. It is not a situation that can simply be blamed on the presence of a phenol.
Many different approaches have been used to correct the negative impression left from discoloration attributed to over-oxidized phenols. One approach is to increase the concentration of the antioxidant system so that the phenol is not put in a position where it is entirely consumed trying to stabilize the polymer. Other representative examples include, but are not limited to:
1) Change the phenolic antioxidant to another that is less prone to discolor, based on the other co-additives, processing conditions, or exposure to oxidizing environments (such as oxides of nitrogen).
2) Incorporate a secondary antioxidant, such as a phosphite, to alleviate the “workload” on the phenolic antioxidant. One can select from a variety of commercially available phosphites, which can have a dramatic effect on the subject of color. Changing the acid scavenger can have a subtle, yet observable effect on the color of the polymer.
As mentioned, the addition of an acid scavenger, e.g., calcium stearate, zinc stearate, zinc oxide, calcium lactate or dihydrotalcite can promote varying degrees of whiteness even though the antioxidant system is held constant. The addition of zinc stearate can be used as an effective way of treating the “symptoms” known as “discoloration.” It should be recognized however, that this approach can be interpreted as treating the “symptom” rather than treating the “disease.”
Again, the key issue here is to determine the root cause of the discoloration and to not simply blame a particular class of molecules for the undesirable discoloration. However, removal of the phenolic antioxidant is an approach that can be used to avoid the discoloration commonly associated with over-oxidized phenols.
3) Change from a phenol based stabilization system to a phenol-free based stabilization system. This approach has been found to be quite useful in many grades of PP and selected grades of PE.
References
Fleck, J. E., “Oven Aging Antioxidant Study to Alleviate Yellowness upon Warehouse Storage,” Internal Technical Report, Ampacet Corp., Terre Haute 1997
Holtzen, D. A., Plastics Engineering, “Discoloration of Pigmented Polyolefins,” 33(4):Reprint (1977)
Holtzen, D. A., Plastics Compounding, “Polyolefin Discoloration,” pp.38-44, November/December 1991
King, R. E., Personal Correspondence, Ciba Additives, 1998
Patel, P.R., Personal Discussions, Ampacet Corp., 1997
Guo, M., Horsey, D., “Stabilization of Agricultural Films: Basics and New Development,” 27th National Agricultural Plastics Congress, Tucson 1998